High resolution NEXAFS of perylene and PTCDI: a surface science approach to molecular orbital analysis†
Abstract
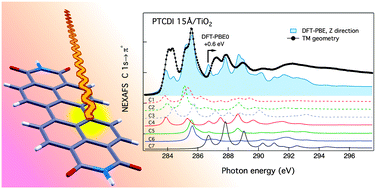
We made use of synchrotron radiation to perform near edge X-ray absorption fine structure spectroscopy, NEXAFS, at the carbon K-edge of perylene and perylene-tetracarboxylic-diimide, PTCDI. Reference spectra measured for isolated molecules in the gas phase are compared with polarization dependent NEXAFS spectra measured on highly oriented thin films in order to study the symmetry of the molecular orbitals. The molecular overlayers are grown onto the rutile TiO2(110) surface for which the large anisotropic corrugation effectively drives the molecular orientation, while its dielectric nature prevents the rehybridization of the molecular orbitals. We employed density functional theory, DFT, calculations to disentangle the contribution of specific carbon atoms to the molecular density of states. Numerical simulations correctly predict the observed NEXAFS azimuthal dichroism of the σ* resonances above the ionization threshold, from which we determine the full geometric orientation of the overlayer molecules. A discrepancy observed for the spectral contribution of the imide carbon atom to the calculated unoccupied molecular orbitals has been explained in terms of initial state effects, as determined by Hartree–Fock corrections and in full agreement with the corresponding shift of the C 1s core level measured by X-ray photoelectron spectroscopy, XPS.

 Please wait while we load your content...
Please wait while we load your content...