Engineering self-assembled N-doped graphene–carbon nanotube composites towards efficient oxygen reduction electrocatalysts†
Abstract
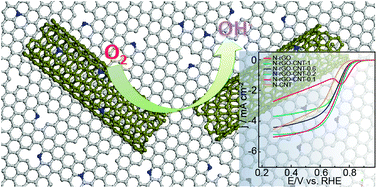
The importance of the oxygen reduction reaction (ORR) in fuel cells and high energy density metal–air batteries has attracted intense research interests in looking for low-cost ORR catalysts as substitutes for expensive and scarce Pt-based catalysts. N-doped graphene and carbon nanotubes prepared in a low-cost and scalable way have demonstrated their potential although the performance still needs to be improved. In view of the requirements for a high-performance ORR electrocatalyst, this work focused on developing the nanocomposites of N-doped reduced graphene oxide (N-rGO) and N-doped carbon nanotubes (N-CNT) as low-cost efficient ORR catalysts by integrating the advantages of abundant highly-active sites from N-rGO and a three-dimensional conductive network for efficient mass and electron transport from N-CNT. By optimizing the preparation method and dedicatedly tuning the composition, the much enhanced ORR activity and superior durability and tolerance to methanol were achieved for the self-assembled N-doped composite (N-rGO–CNT) at a mass ratio of 1 : 5 rGO/CNT. Further improvement of the ORR electrocatalytic activity of the composite was also demonstrated by introducing iron into the composite.
- This article is part of the themed collection: Electrocatalysis - Fundamental Insights for Sustainable Energy

 Please wait while we load your content...
Please wait while we load your content...