Time-resolved fluorescence spectroscopy study of excited state dynamics of alkyl- and benzo-substituted triphyrin(2.1.1)
Abstract
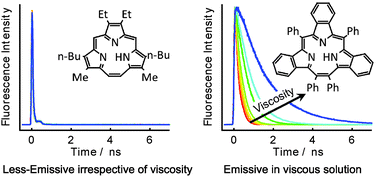
We have investigated the photophysical properties of alkyl-substituted triphyrin(2.1.1) (ATp) and benzotriphyrin(2.1.1) (BTp) by steady-state and time-resolved fluorescence spectroscopy. We focused on the effect of NH proton tautomerization, planarity of the macrocycles, and substituents on these properties. The fluorescence quantum yields (Φy) of ATp did not depend on solvent viscosity, whereas those of BTp increased with solvent viscosity, reaching a maximum value of 0.17 in paraffin. Interestingly, analyzing Φy showed that the non-radiative rate constant of BTp decreased sharply as the solvent viscosity increased. These results suggest that the substituted phenyl groups play a crucial role in suppressing molecular distortion, thus leading to decreased non-radiative relaxation in triphyrin(2.1.1). The hydrogen bond formed in the inner cavity potentially contributes to the suppression of the structural distortion, whereas the pyrrole rings in the macrocycle are close, as in porphycene.

 Please wait while we load your content...
Please wait while we load your content...