Mechanistic insight into highly efficient gas permeation and separation in a shape-persistent ladder polymer membrane†
Abstract
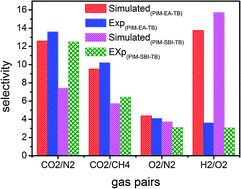
A fully atomistic simulation study is reported to provide mechanistic insight into the superior performance experimentally observed for a polymer membrane (Carta et al., Science, 2013, 339, 303–307). The membrane namely PIM-EA-TB is produced by a shape-persistent ladder polymer of intrinsic microporosity (PIM) with rigid bridged bicyclic ethanoanthracene (EA) and Tröger's base (TB). The simulation reveals that PIM-EA-TB possesses a larger surface area, a higher fraction free volume and a narrower distribution of torsional angles compared to PIM-SBI-TB, which consists of less rigid spirobisindane (SBI). The predicted surface areas of PIM-EA-TB and PIM-SBI-TB are 1168 and 746 m2 g−1, close to experimental values of 1120 and 745 m2 g−1, respectively. For five gases (CO2, CH4, O2, N2 and H2), the solubility and diffusion coefficients from simulation match well with experimental data, except for H2. The solubility coefficients decrease in the order of CO2 > CH4 > O2 > N2 > H2, while the diffusion coefficients increase following CH4 < CO2 < N2 < O2 < H2. In terms of the separation for CO2/N2, CO2/CH4 and O2/N2 gas pairs, PIM-EA-TB exhibits higher permselectivities than PIM-SBI-TA, in good agreement with experiment. From a microscopic perspective, this simulation study elucidates that the presence of bridged bicyclic units in PIM-EA-TB enhances the rigidity of polymer chains as well as the capability of gas permeation and separation, and the bottom-up insight could facilitate the rational design of new high-performance membranes.

 Please wait while we load your content...
Please wait while we load your content...