A photo- and electrochemical investigation of BODIPY–cobaloxime complexes for hydrogen production, coupled with quantum chemical calculations†
Abstract
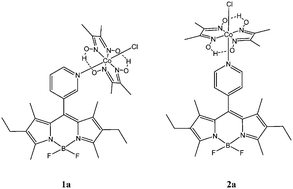
Two BODIPY–cobaloxime complexes; [{Co(dmgH)2Cl}{3-[bis-(4-ethyl-3,5-dimethyl-1H-pyrrol-2-yl)-methyl]-pyridine-borondiflouride}] (1a) and [{Co(dmgH)2Cl}{4-[bis-(4-ethyl-3,5-dimethyl-1H-pyrrol-2-yl)-methyl]-pyridine-borondiflouride}] (2a) (BODIPY = boron dipyrromethene), (dmgH = dimethylglyoxime) have been synthesised and studied as model catalytic systems for the generation of hydrogen gas in aqueous media. Under photochemical conditions, neither complex catalysed the reduction of water to hydrogen. However, both complexes showed considerable activity under electrochemical conditions. Turn-over-numbers for hydrogen production of 1.65 × 104 and 1.08 × 104 were obtained for 1a and 2a respectively following potentiostatic electrolysis at −1.2 V vs. Ag/AgCl after 1 hour. Quantum chemical calculations were performed to provide an explanation for the lack of photochemical activity.

 Please wait while we load your content...
Please wait while we load your content...