Iron based photoanodes for solar fuel production
Abstract
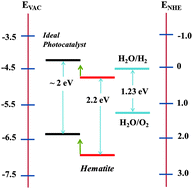
In natural photosynthesis, the water splitting reaction of photosystem II is the source of the electrons/reducing equivalents for the reduction of carbon dioxide to carbohydrate while oxygen is formed as the by-product. Similarly, for artificial photosynthesis where the end product is a solar fuel such as hydrogen, a water splitting-oxygen evolving system is required to supply high energy electrons to drive the reductive reactions. Very attractive candidates for this purpose are iron based semiconductors which have band gaps corresponding to visible light and valence band energies sufficient to oxidise water. The most studied system is hematite (Fe2O3) which is highly abundant with many attributes for incorporation into photoelectrochemical (PEC) cells. We review the recent progress in manipulating hematite for this purpose through nanostructuring, doping and surface modifications. We also consider several hybrid iron-based semiconducting systems like ferrites and iron titanates as alternatives to hematite for light driven water splitting emphasizing their advantages with respect to their band levels and charge transport properties.
- This article is part of the themed collection: Photosynthesis: From Natural to Artificial

 Please wait while we load your content...
Please wait while we load your content...