High activity of cubic PtRh alloys supported on graphene towards ethanol electrooxidation
Abstract
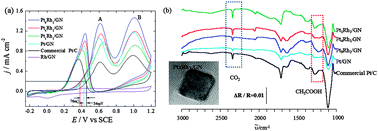
Cubic PtRh alloys supported on graphene (PtxRhy/GN) with different atomic ratio of Pt and Rh were directly synthesized for the first time using the modified polyol method with Br− for the shape-directing agents. The process didn't use surface-capping agents such as PVP that easily occupy the active sites of electrocatalysts and are difficult to remove. Graphene is the key factor for cubic shape besides Br− and keeping catalysts high-dispersed. The X-ray diffraction (XRD), scanning electron microscope (SEM) and transmission electron microscope (TEM) were used to characterize the structure and morphology of these electrocatalysts. The results showed that they were composed of homogeneous cubic PtRh alloys. Traditional electrochemical methods, such as cyclic voltammetry and chronoamperometry, were used to investigate the electrocatalytic properties of PtxRhy/GN towards ethanol electrooxidation. It can be seen that PtxRhy/GN with all atomic ratios exhibited high catalytic activity, and the most active one has a composition with Pt : Rh = 9 : 1 atomic ratio. Electrochemical in situ FTIR spectroscopy was used to evaluate the cleavage of C–C bond in ethanol at room temperature in acidic solutions, the results illustrated that Rh in an alloy can promote the split of C–C bond in ethanol, and the alloy catalyst with atomic ratio Pt : Rh = 1 : 1 showed obviously better performance for the C–C bond breaking in ethanol and higher selectivity for the enhanced activity of ethanol complete oxidation to CO2 than alloys with other ratios of Pt and Rh. The investigation indicates that high activity of PtxRhy/GN electrocatalyst towards ethanol oxidation is due to the specific shape of alloys and the synergistic effect of two metal elements as well as graphene support.
- This article is part of the themed collection: Electrocatalysis - Fundamental Insights for Sustainable Energy

 Please wait while we load your content...
Please wait while we load your content...