Solid state chemistry of nitrogen oxides – Part II: surface consumption of NO2
Abstract
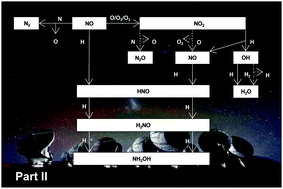
Nitrogen oxides are considered to be important astrochemical precursors of complex species and prebiotics. However, apart from the hydrogenation of solid NO that leads to the surface formation of hydroxylamine, little is known about the full solid state reaction network involving both nitrogen and oxygen. Our study is divided into two papers, hereby called Part I and Part II. In the accompanying paper, we investigate the surface reactions NO + O/O2/O3 and NO + N with a focus on the formation of NO2 ice. Here, we complement this study by measurements of the surface destruction of solid NO2, e.g., NO2 + H/O/N. Experiments are performed in two separate ultra-high vacuum setups and therefore under different experimental conditions to better constrain the experimental results. Surface reaction products are monitored by means of Fourier Transform Reflection Absorption Infrared Spectroscopy (FT-RAIRS) and Temperature Programmed Desorption (TPD) techniques using mass spectrometry. The surface destruction of solid NO2 leads to the formation of a series of nitrogen oxides such as NO, N2O, N2O3, and N2O4 as well as HNO, NH2OH, and H2O. When NO2 is mixed with an interstellar more relevant apolar (i.e., CO) ice, solid CO2 and HCOOH are also formed due to interactions between different reaction routes. The astrophysical implications of the full nitrogen and oxygen reaction network derived from Parts I and II are discussed.

 Please wait while we load your content...
Please wait while we load your content...