An inter-tangled network of redox-active and conducting polymers as a cathode for ultrafast rechargeable batteries
Abstract
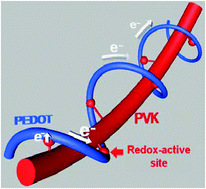
A 1D organic redox-active material is combined with another 1D conductive material for rechargeable batteries. Poly(vinyl carbazole) (or PVK) and poly(3,4-ethylenedioxythiophene) doped with poly(styrenesulfonate) (or PEDOT:PSS) are used as the redox-active and conductive 1D materials, respectively. Due to their extremely anisotropic geometry, the two polymers are expected to be inter-tangled with each other, showing a kinetically ideal model system in which each redox-active moiety of PVK is supposed to be directly connected with the conducting pathways of PEDOT:PSS. In addition to its role as a conductive agent providing kinetic benefits, PEDOT:PSS works as an efficient binder that guarantees enhanced electrochemical performances with only a tenth of the amount of a conventional binder (polyvinylidene fluoride or PVdF). The benefit of gravimetric energy density gain obtained using the conductive binder comes mainly from efficient spatial coverage of binding volume due to the low density of PEDOT:PSS. Towards realizing flexible all-polymer batteries, a quasi-all-polymer battery half-cell is designed using the PVK/PEDOT:PSS composite with a polymer gel electrolyte.

 Please wait while we load your content...
Please wait while we load your content...