The benzoic acid–water complex: a potential atmospheric nucleation precursor studied using microwave spectroscopy and ab initio calculations†
Abstract
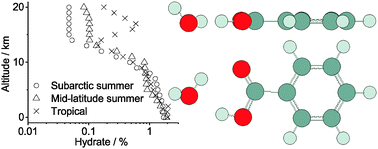
The pure rotational, high-resolution spectrum of the benzoic acid–water complex was measured in the range of 4–14 GHz, using a cavity-based molecular beam Fourier-transform microwave spectrometer. In all, 40 a-type transitions and 2 b-type transitions were measured for benzoic acid–water, and 12 a-type transitions were measured for benzoic acid–D2O. The equilibrium geometry of benzoic acid–water was determined with ab initio calculations, at the B3LYP, M06-2X, and MP2 levels of theory, with the 6-311++G(2df,2pd) basis set. The experimental rotational spectrum is most consistent with the B3LYP-predicted geometry. Narrow splittings were observed in the b-type transitions, and possible tunnelling motions were investigated using the B3LYP/6-311++G(d,p) level of theory. Rotation of the water moiety about the lone electron pair hydrogen-bonded to benzoic acid, across a barrier of 7.0 kJ mol−1, is the most likely cause for the splitting. Wagging of the unbound hydrogen atom of water is barrier-less, and this large amplitude motion results in the absence of c-type transitions. The interaction and spectroscopic dissociation energies calculated using B3LYP and MP2 are in good agreement, but those calculated using M06-2X indicate excess stabilization, possibly due to dispersive interactions being over-estimated. The equilibrium constant of hydration was calculated by statistical thermodynamics, using ab initio results and the experimental rotational constants. This allowed us to estimate the changes in percentage of hydrated benzoic acid with variations in the altitude, region, and season. Using monitoring data from Calgary, Alberta, and the MP2-predicted dissociation energy, a yearly average of 1% of benzoic acid is expected to be present in the form of benzoic acid–water. However, this percentage depends sensitively on the dissociation energy. For example, when using the M06-2X-predicted dissociation energy, we find it increases to 18%.

 Please wait while we load your content...
Please wait while we load your content...