Coupling of conformational and ionization equilibria in linear poly(ethylenimine): a study based on the site binding/rotational isomeric state (SBRIS) model
Abstract
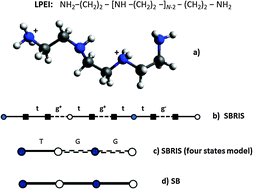
The conformational and ionization properties of linear poly(ethylenimine) are studied by combining the site binding model (SB) with the rotational isomeric state (RIS) model, developed by Flory to calculate the conformational properties of neutral linear molecules. The resulting approach (the SBRIS model) is used to rationalise the experimental poly(ethylenimine) titration curves. By fitting the experimental macroconstants, conformational and binding parameters are obtained. The obtained values are consistent with previous binding and structural information. The emergence of triplet interaction between protonated sites is explained as a natural consequence of the coupling between binding and conformational equilibria. When only nearest neighbour interactions are considered, transfer matrix techniques are used in the calculations. In order to account for excluded volume and long-range electrostatic interactions, Monte Carlo simulations are performed. The results indicate that at high ionic strengths, long-range interactions have a very limited impact on the titration curves. However, for long chains, they have a significant influence on the radius of gyration.

 Please wait while we load your content...
Please wait while we load your content...