Room temperature light-induced recrystallization of Cu2O cubes to CuO nanostructures in water†
Abstract
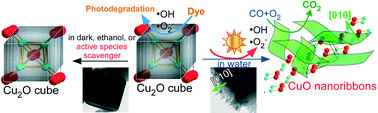
The mechanism of temperature-assisted Cu and Cu2O oxidation in oxygen and subsequent CuO nanowire growth is well known. Here, we report a simple light-induced process for the recrystallization of Cu2O cubes to [010] growth-directed CuO nanoribbons in water at room temperature. This was attributed to the formation and participation of ˙OH and ˙O2− active species in water as well as the outward diffusion of Cu ions leading to the formation of CuO nanoribbons along unstable {010} facets on the surface of Cu2O cubes. The oxidation was significantly suppressed under dark conditions or by active species scavengers, confirming the strong role of visible light. CO oxidation activity of nanoribbons was found to be superior to that of cubes. Our unique light-induced recrystallization of Cu2O to CuO in water provides new insight and better understanding of the oxidation mechanism of Cu2O, enabling tailoring of nanostructures by varying types of light (sun, incandescent and fluorescent) and opening a new strategy for development of energy- and environment-related Cu oxide nanomaterials.

 Please wait while we load your content...
Please wait while we load your content...