Crystal form selectivity by humidity control: the case of the ionic co-crystals of nicotinamide and CaCl2†
Abstract
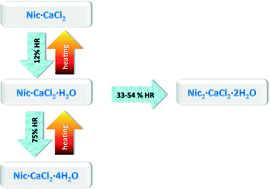
Post-synthesis (de)hydration techniques were used here to explore further hydrated forms of ionic co-crystals (ICCs) of nicotinamide with CaCl2. Humidity is shown to be a crucial factor for ICCs, which cannot be ignored for a complete polymorph screening of this class of compounds. The exposure of nicotinamide·CaCl2·H2O obtained by kneading reaction to a controlled relative humidity of 75% led to the formation of a new hydrated phase: nicotinamide·CaCl2·4H2O. When nicotinamide·CaCl2·H2O was exposed to a relative humidity between 32% and 54%, nicotinamide2·CaCl2·2H2O was obtained. The anhydrous form was achieved as the result of overnight dehydration of nicotinamide·CaCl2·H2O at 150 °C. The tetrahydrate form cannot be obtained as a first product for kinetic (or thermodynamic) reasons. Control of the relative humidity has proven to be an efficient way to selectively isolate and stabilize powders of pure hydrous ICC phases, which is fundamental for industrial applications. Crystalline structures of nicotinamide·CaCl2·4H2O and anhydrous nicotinamide·CaCl2 were determined by powder diffraction.
- This article is part of the themed collection: Functional Co-crystals

 Please wait while we load your content...
Please wait while we load your content...