Influence of the pore structure of CeO2 supports on the surface texture and catalytic activity for CO oxidation
Abstract
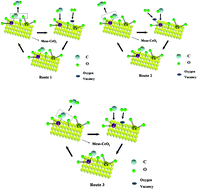
In this paper, three kinds of CeO2 nano-materials with different pore structures, i.e., mesoporous, microporous and nanoparticle CeO2, were synthesized. Mesoporous CeO2 (meso-CeO2) and microporous CeO2 (micro-CeO2) were prepared by adopting the mesoporous silica KIT-6 and microporous high silica ZSM-5 (Si/Al = 344.1) as templates, respectively. CeO2 nanoparticles (nano-CeO2) were synthesized by precipitation method. The palladium loaded meso-CeO2, micro-CeO2 and nano-CeO2 supports were evaluated for their catalytic activity in the CO oxidation reaction. The Pd/meso-CeO2 exhibited the highest catalytic activity, and the complete conversion temperature (T100) was about 50 °C for the CO oxidation. According to the analysis, the meso-CeO2 support has a mesoporous structure, large BET surface area and small particle size. At the same time, the Pd/meso-CeO2 catalyst has a large number of active surface oxygen species, Ce3+ cationic species and Pd4+ cationic species. These above characteristics of Pd/meso-CeO2 were relatively conducive to the catalytic oxidation of CO.

 Please wait while we load your content...
Please wait while we load your content...