Positions of amino groups on ammonium salts tunes the conformations of crown ethers: crystal structures, Hirshfeld surfaces and spectroscopic studies†
Abstract
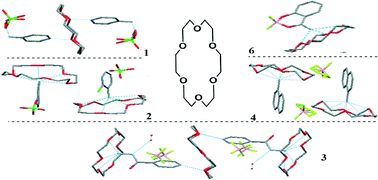
Six supramolecular assembly complexes: pyridine-4-ammonium-18-crown-6-perchlorate ([(C5H7 N2)2·(18-C-6)]2+·2(ClO4)−, 1), 2-chloride-pyridine-3-ammonium-18-crown-6-perchlorate ([(C5H6N2Cl)·(18-C-6)]+·(ClO4)−, 2), isoniazide-ammonium-18-crown-6-tetrafluoroborate ([(C6H9N3O)2·(18-C-6)3]4+·4(BF4)−·4(H2O), 3), naphthylammonium-18-crown-6-tetrafluoroborate ([(C10H10N)·(18-C-6)]+·(BF4)−, 4), benzidine-ammonium-15-crown-5-tetrafluoroborate ([(C12H14N2)·(15-C-5)2]2+·2(BF4)−, 5), salicylamide-18-crown-6-tetrafluoroborate ([(C7H6NO2BF2)·(18-C-6)], 6) have been prepared and investigated in detail by single-crystal X-ray diffraction, Hirshfeld surfaces analysis, Raman and IR spectroscopy. The position of the amino group on the ammonium salt plays a key role in the supramolecular structure and conformation of the crown ethers: 1 and 3 formed five- and nine-membered pendulum-like structures, respectively; 2 and 4 formed umbrella-like structures; 5 formed a “dumbbell-like” structure with 15-crown-5; the salicylamide in 6 formed a novel complex with tetrafluoroboric acid. The 18-crown-6 molecule displays a “round” “D3d like” conformation in 1 and 6, while it displays a “boat-form” in 2 and 4, and two different conformations in 3 with “round” “D3d like” and “chair-like” conformations in a stoichiometric ratio of 2 : 1. The differences in conformation has a significant effect on the intermolecular interactions and spectroscopic properties: the “D3d like” is more apt to H–H and O–H contacts than the “chair-like”, while the latter is apt to C–H⋯π contacts. In the presence of ammonium salts, the characteristic Raman peaks at 418 cm−1 and within 900–800 cm−1 are maintained for the “D3d like” conformation, while they vanish for the “boat-form” and “chair-like”. In addition, while the characteristic IR peak within the 1130–1090 cm−1 region was narrowed for the “D3d like”, it was broadened for the other two conformations.

 Please wait while we load your content...
Please wait while we load your content...