Enhancing the crystallization of poly(l-lactide) using a montmorillonitic substrate favoring nucleation
Abstract
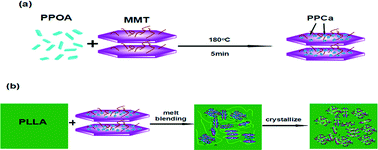
Montmorillonite (MMT) generally has weak nucleating ability or even retarded crystallization for poly(L-lactide) (PLLA) depending on the dispersion morphology in the matrix. A novel MMT with a nucleating surface (NMMT) chemically supported by calcium phenylphosphonic acid (PPCa) (an effective nucleant of PLLA), was prepared through the chemical reaction between phenylphosphonic acid (PPOA) and a calcium ion on the surface of MMT for the first time. Differential scanning calorimetry, infrared spectroscopy and wide-angle X-ray diffraction confirm the reaction between PPOA and Ca-montmorillonite and the formation of PPCa on the surface of MMT. Then, NMMT was introduced into PLLA via simple melt blending. The most intriguing result is that the crystallization rate of PLLA greatly increases after incorporation of NMMT, and the crystallization rate of PLLA increases with increasing NMMT fraction and decreasing MMT/PPOA mass ratio. The nucleation density of PLLA increases and the spherulite size decreases significantly in the presence of NMMT. Epitaxy is the possible mechanism to explain the nucleation phenomenon of the PLLA/NMMT system. Moreover, the tensile test results show that NMMT has a strengthening effect on the amorphous PLLA. Through a short time annealing procedure, the mechanical properties such as the tensile modulus and storage modulus of PLLA are improved by the addition of NMMT.

 Please wait while we load your content...
Please wait while we load your content...