Variation of CO2 adsorption in isostructural Cd(ii)/Co(ii) based MOFs by anion modulation†
Abstract
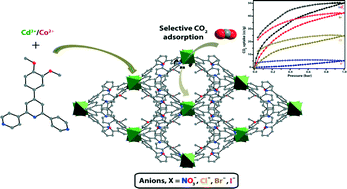
A potential multidentate tris-pyridyl ligand has been synthesized, and later employed for the tailoring of four isostructural cadmium(II)/cobalt(II) two dimensional (2D) metal organic frameworks, [[M(dmpt)2(X)2]·xS]n, where S = solvent, M = Cd2+, (X = NO3− (1), Br− (2), I− (3)); M = Co2+ (X = Cl− (4)), dmpt = 4′-(3,4-dimethoxyphenyl)-4,2′:6′,4′′-terpyridine. These complexes have been characterized by infrared (IR) spectroscopy, elemental analysis, thermogravimetric analysis (TGA) and powder X-ray diffraction (PXRD) and single crystal X-ray diffraction analyses. Single crystal X-ray diffraction analysis reveals that the complexes (1–4) are isostructural and crystallize in the monoclinic C2/c space group. They possess similar structural features such as metal-coordination environment, network topology, π⋯π interactions and exhibit a 2D uninodal 44-sql topology with slightly tuned cavities. In complexes 1–4, the [[M(dmpt)2(X)2]·xS] coordination monomer acts as the building block, building 2D layers which are further stacked up via Van der Waals interactions into a three-dimensional (3D) framework. The effect of weak interactions on the structural dynamics of the systems has also been studied. Furthermore, a gas adsorption study of these complexes demonstrates that they are affected by the impact of directing anions inside the pores.

 Please wait while we load your content...
Please wait while we load your content...