Mechanochemical dehydrochlorination and chelation reaction in the solid state: from a molecular salt to a coordination complex†
Abstract
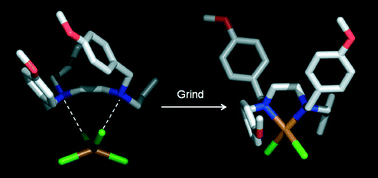
We report the solid state structural transformation of a hydrogen bonded complex salt into a metal complex via dehydrochlorination using mechanochemistry. A crystalline salt containing a large and flexible bidentate dication hydrogen bonded to a tetrachlorometalate (II) anion has been ground in the presence of KOH. Substitution of charge-assisted hydrogen bonding interactions by coordination bonds via chelation has been demonstrated by single-crystal and powder X-ray diffraction analysis. By-product water molecules are included in the structure, playing an important role establishing electrostatic interactions. The irreversibility property of the transformation of the coordination complex into a hydrogen bonded complex salt was determined experimentally. Density functional calculations were used to attempt a rationalisation of the structural results into the mechanochemical reactions.
- This article is part of the themed collection: Editor’s Collection: Mechanochemistry

 Please wait while we load your content...
Please wait while we load your content...