One-pot hydrazide-based native chemical ligation for efficient chemical synthesis and structure determination of toxin Mambalgin-1†
Abstract
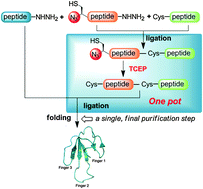
An efficient one-pot chemical synthesis of snake venom toxin Mambalgin-1 was achieved using an azide-switch strategy combined with hydrazide-based native chemical ligation. Synthetic Mambalgin-1 exhibited a well-defined structure after sequential folding in vitro. NMR spectroscopy revealed a three-finger toxin family structure, and the synthetic toxin inhibited human acid-sensing ion channel 1a.

 Please wait while we load your content...
Please wait while we load your content...