Paracrine signalling of inflammatory cytokines from an in vitro blood brain barrier model upon exposure to polymeric nanoparticles†
Abstract
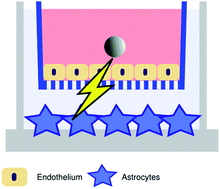
Nanoparticle properties, such as small size relative to large highly modifiable surface area, offer great promise for neuro-therapeutics and nanodiagnostics. A fundamental understanding and control of how nanoparticles interact with the blood-brain barrier (BBB) could enable major developments in nanomedical treatment of previously intractable neurological disorders, and help ensure that nanoparticles not intended to reach the brain do not cause adverse effects. Nanosafety is of utmost importance to this field. However, a distinct lack of knowledge exists regarding nanoparticle accumulation within the BBB and the biological effects this may induce on neighbouring cells of the Central Nervous System (CNS), particularly in the long-term. This study focussed on the exposure of an in vitro BBB model to model carboxylated polystyrene nanoparticles (PS COOH NPs), as these nanoparticles are well characterised for in vitro experimentation and have been reported as non-toxic in many biological settings. TEM imaging showed accumulation but not degradation of 100 nm PS COOH NPs within the lysosomes of the in vitro BBB over time. Cytokine secretion analysis from the in vitro BBB post 24 h 100 nm PS COOH NP exposure showed a low level of pro-inflammatory RANTES protein secretion compared to control. In contrast, 24 h exposure of the in vitro BBB endothelium to 100 nm PS COOH NPs in the presence of underlying astrocytes caused a significant increase in pro-survival signalling. In conclusion, the tantalising possibilities of nanomedicine must be balanced by cautious studies into the possible long-term toxicity caused by accumulation of known ‘toxic’ and ‘non-toxic’ nanoparticles, as general toxicity assays may be disguising significant signalling regulation during long-term accumulation.
- This article is part of the themed collection: Analytical Toxicology of Nanoparticles

 Please wait while we load your content...
Please wait while we load your content...