Statistical evaluation of toxicological bioassays – a review
Abstract
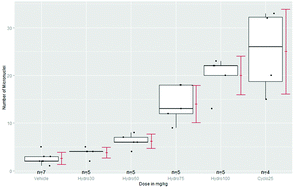
The basic conclusions in almost all reports on new drug applications and in all publications in toxicology are based on statistical methods. However, serious contradictions exist in practice: designs with small samples sizes but use of asymptotic methods (i.e. constructed for larger sample sizes), statistically significant findings without biological relevance (and vice versa), proof of hazard vs. proof of safety, testing (e.g. no observed effect level) vs. estimation (e.g. benchmark dose), available statistical theory vs. related user-friendly software. In this review the biostatistical developments since about the year 2000 onwards are discussed, mainly structured for repeated-dose studies, mutagenicity, carcinogenicity, reproductive and ecotoxicological assays. A critical discussion is included on the unnecessarily conservative evaluation proposed in guidelines, the inadequate but almost always used proof of hazard approach, and the limitation of data-dependent decision-tree approaches.

 Please wait while we load your content...
Please wait while we load your content...