Multifunctional micro- and nanosized metal–organic frameworks assembled from bisphosphonates and lanthanides†
Abstract
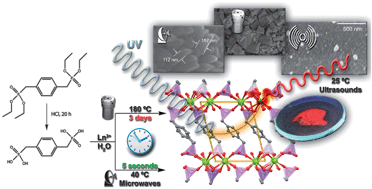
Phase-pure and highly crystalline [Ln(Hpmd)(H2O)] materials [where Ln3+ = Eu3+ (1), Gd3+ (2) and Tb3+ (3); H4pmd = 1,4-phenylenebis(methylene)diphosphonic acid] were prepared by using three distinct approaches: (i) conventional hydrothermal synthesis (180 °C, 3 days); (ii) microwave-assisted heating (50 W irradiation power, 40 °C, 5 seconds; nano-sized aggregates with sizes ranging between 100 and 150 nm); and (iii) ultrasound-assisted synthesis which, for highly diluted reactive mixtures (Ln3+ : H4pmd : H2O of 1 : 1 : 7200), permitted the preparation of isolated nano-crystals at ambient temperature with 5 minutes of ultrasonic irradiation. Compounds were structurally characterized by powder X-ray diffraction (Rietveld refinement and variable-temperature studies), thermogravimetry, vibrational spectroscopy, elemental analysis and electron microscopy (SEM and EDS). The magnetic behavior of 1 and 2 was investigated between ambient temperature and ca. 2 K revealing that the Ln3+ cations act as isolated centers. A strategy to calculate the vibrational spectra of MOF structures and based on the known embedded-clusters approach is proposed and applied to [Ln(Hpmd)(H2O)]. This allowed the identification, along with deuteration of the materials, of the vibrational modes of the confined water molecule in the structure. Compound 1 was tested in the methanolysis of styrene oxide at 55 °C: it is shown that microcrystalline 1 (1-m) does not possess significant catalytic activity; on the other hand, the nano-sized counterpart (1-n) exhibits relatively high catalytic activity and excellent selectivity to 2-methoxy-2-phenylethanol (100% yield within 48 h of reaction time). Photoluminescence studies both at ambient and low temperatures showed, on the one hand, that bulk materials are composed of a single lanthanide site and, on the other hand, that the organic linker is a suitable sensitizer of Tb3+ (absolute quantum yield of ca. 14% for 3). It is further demonstrated that the coordinated water molecule has a pivotal role in the quenching of the photoluminescence of Eu3+ in 1: deuteration of the material results in a ca. 3.4 times improvement of the decay time (e.g., at 300 K the lifetime improves from 0.58 ± 0.01 ms to 1.98 ± 0.01 ms).
- This article is part of the themed collection: Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...