Conjugated polymers with m-pyridine linkages: synthesis, photophysics, solution structure and film morphology†
Abstract
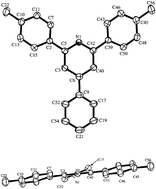
The synthesis of two π-conjugated polymers whose structures include m-pyridyl linkages that create regularly spaced bends and disrupt π-conjugation along the polymer backbones is described. The π-conjugated segments in the two polymers were comprised of p-(2,5-dialkoxyphenylene)vinylene, p-phenylenevinylene and 2,7-carbazole subunits. Additionally, each m-pyridylene unit is enshrouded within a steric cleft provided by 2,6-diaryl substitution of the pyridyl ring, thus minimizing inter-chain interactions of these sites. The concentration-dependence of photophysical characteristics along with small angle neutron scattering (SANS) was employed to resolve solution aggregation properties and atomic force microscopy was employed to assess the surface morphology of polymer films.

 Please wait while we load your content...
Please wait while we load your content...