Development of an efficient transdermal drug delivery system with TAT-conjugated cationic polymeric lipid vesicles
Abstract
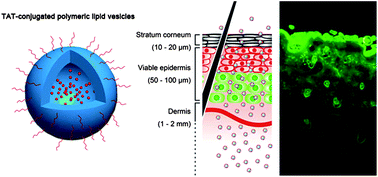
Conventional liposomes (CLs) have been used as a transdermal drug delivery system for enhancing the delivery of hydrophilic drugs into/through the skin. However, their applications have been constrained by their limited penetration ability and poor stability. In this article, a new kind of transactivating transcriptional activator peptide (TAT)-conjugated polymeric lipid vesicles (TPLVs) formed from amphiphilic lysine–linoleic acid modified dextran (LLD) and cholesterol (Chol) has been prepared successfully. The newly developed TPLVs had a bilayer structure similar to CLs. The TPLVs also have smaller particle size, narrower distribution, higher positive charge and much better stability than the CLs; they remained stable in aqueous solutions for up to 60 days without aggregation. The in vitro and in vivo skin permeation studies revealed that TPLVs delivered a higher amount of drug through the skin than CLs, indicating enhanced drug transdermal activities. The synergetic effects of abovementioned features and the cell-penetrating peptide TAT might have contributed to the improved skin penetration ability of the TPLVs. Similar to CLs, TPLVs began to show limited cytotoxicity against human umbilical vein endothelial cells at a concentration of 200 μg mL−1. The in vitro release profiles showed that the TPLVs achieved a sustained release of lidocaine. These results suggest that the TPLVs may be utilized as an efficient carrier to replace CLs for transdermal drug delivery.

 Please wait while we load your content...
Please wait while we load your content...