Supramolecular assembly of polythiophene-g-polymethacrylic acid-doped polyaniline with interesting morphological and opto-electronic properties†
Abstract
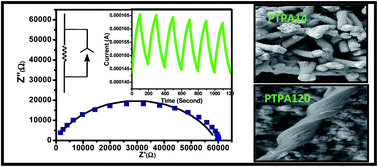
Polythiophene-g-poly(methacrylic acid) (PTMA) is prepared by atom transfer radical polymerization (ATRP) of tert-butyl methacrylate upon a polythiophene backbone followed by hydrolysis of tert-butyl groups using trifluoroacetic acid. PTMA is characterised by 1H NMR and gel permeation chromatography (GPC). It acts as both a template and a dopant for the synthesis of polyaniline (PANI) nanostructures, and the PTMA concentration is varied to tune the physical properties of PTMA–PANI (PTPA) hybrids. Field-emission scanning electron microscopy (FESEM) exhibits the formation of nanorod morphology till 1 : 10 composition (PTMA–PANI; w/w) and for the PTPA120 hybrids helical nanorods are produced along with some small size spheroids. UV-vis spectra of the PTPA hybrids indicate the formation of PTMA-doped PANI; moreover, with increase in the concentration of dopant PTMA, the π band to the polaron band transition peak is red-shifted. Circular dichroism (CD) spectra of the hybrid exhibit a broad positive peak at 310–470 nm, indicating the presence of single-handed chirality in all the PTPA hybrids. Fourier transformed infrared (FTIR) spectra also confirm the doped nature of PANI in the PTPA hybrids, and X-ray diffraction (XRD) data indicates that there is self-organization of PTMA and PANI due to π–π interaction. Thermogravimetric analysis data indicate an increase of thermal stability of PTPA hybrids compared to that of PTMA. The PTPA hybrids are semiconducting in nature (dc-conductivity 10−2 to 10−5 s cm−1), and they exhibit reproducible photo-conductivity by alternate “On” and “Off” switching of white light illumination. Analysis of impedance results suggests the presence of an equivalent circuit with a capacitance value of 3 × 10−10 F and a bulk resistance of 5.8 × 104 Ω.

 Please wait while we load your content...
Please wait while we load your content...