Multiple phases in the ε-VPO4O–LiVPO4O–Li2VPO4O system: a combined solid state electrochemistry and diffraction structural study†
Abstract
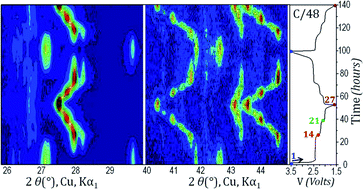
Polyanionic materials attract great interest in the field of Li-ion battery research thanks to the wide range of possible available compositions, resulting in a great amount of different properties. Tavorite type compositions offer a very rich crystal chemistry, among which LiVPO4F delivers the highest theoretical energy density. In this work we focus our interest on the homeotypic composition LiVPO4O. This oxy-phosphate shows the ability to exploit two redox couples, V5+/V4+ at 3.95 V vs. Li+/Li0 and V4+/V3+ at an average potential of 2.3 V vs. Li+/Li0 upon Li+ extraction and insertion, respectively. The two domains show marked differences both in the electrochemical signature and in the phase diagram. Here we address for the first time both topics with a combination of electrochemical techniques and ex situ/in situ X-ray and neutron diffraction and support of density functional theory (DFT) calculations, to get a deep understanding of the behavior of Li1±xVPO4O. In the low voltage region, in particular, the formation of intermediate phases and the crystal structure of the end member Li2VPO4O are reported for the first time.

 Please wait while we load your content...
Please wait while we load your content...