Strontium influence on the oxygen electrocatalysis of La2−xSrxNiO4±δ (0.0 ≤ xSr ≤ 1.0) thin films†
Abstract
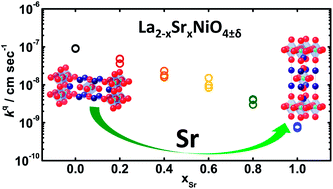
Substitution of lanthanum by strontium (Sr) in the A-site of cobalt-containing perovskites can greatly promote oxygen surface exchange kinetics at elevated temperatures. Little is known about the effect of A-site substitution on the oxygen electrocatalysis of Ruddlesden–Popper (RP) oxides. In this study, we report, for the first time, the growth and oxygen surface exchange kinetics of La2−xSrxNiO4±δ (LSNO, 0.0 ≤ xSr ≤ 1.0) thin films grown on (001)cubic-Y2O3-stabilized ZrO2 (YSZ) by pulsed laser deposition. High-resolution X-ray diffraction analysis revealed that the LSNO film orientation was changed gradually from the (100)tetra. (in-plane) to the (001)tetra. (out-of-plane) orientation in the RP structure with increasing Sr from La2NiO4+δ (xSr = 0) to LaSrNiO4±δ (xSr = 1.0). Such a change in the LSNO film orientation was accompanied by reduction in the oxygen surface exchange kinetics by two orders of magnitude as shown from electrochemical impedance spectroscopy results. Density functional theory (DFT) calculations showed that Sr substitution could stabilize the (001)tetra. surface relative to the (100)tetra. surface and both Sr substitution and increasing (001)tetra. surface could greatly weaken adsorption of molecular oxygen in the La–La bridge sites in the RP structure, which can reduce oxygen surface exchange kinetics.

 Please wait while we load your content...
Please wait while we load your content...