Förster resonance energy transfer confirms the bacterial-induced conformational transition in highly-branched poly(N-isopropyl acrylamide with vancomycin end groups on binding to Staphylococcus aureus
Abstract
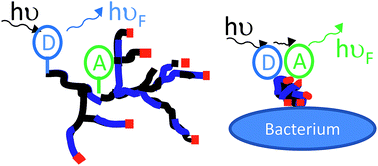
We describe a series of experiments designed to investigate the conformational transition that highly-branched polymers with ligands undergo when interacting with bacteria, a process that may provide a new sensing mechanism for bacterial detection. Fluorescent highly-branched poly(N-isopropyl acrylamide)s (HB-PNIPAM) were prepared by sequential self-condensing radical copolymerizations, using anthrylmethyl methacrylate (AMMA) and fluorescein-O-acrylate (FA) as fluorescent comonomers and 4-vinylbenzyl pyrrole carbodithioate as a branch forming monomer. Differences in reactivity necessitated to first copolymerize AMMA then react with FA in a separate sequential monomer feed step. Modifications of the chain ends produced vancomycin-functional derivatives (HB-PNIPAM-Van). The AMMA and FA labels allow probing of the conformational behaviour of the polymers in solution via Förster resonance energy transfer experiments. It was shown that interaction of this polymer's end groups with Staphylococcus aureus induced a macromolecular collapse. The data thus provide conclusive evidence for a conformational transition that is driven by binding to a bacterium.

 Please wait while we load your content...
Please wait while we load your content...