Ultrafast and reversible thermochromism of a conjugated polymer material based on the assembly of peptide amphiphiles†‡
Abstract
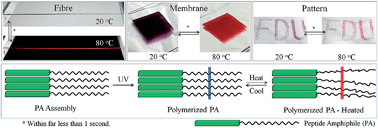
The translation of the colour change that occurs in chameleons into synthetic functional materials has attracted increasing interest, and conjugated polymers such as polydiacetylenes that undergo chromatic transitions under heating represent one of the most studied systems. However, they suffer from a slow and irreversible colour change, typically at low temperatures, e.g., 50–90 °C, which limits their sensing applications. In addition, they occur as powders or films and cannot satisfy the requirements for wearable electronics, a mainstream direction in sensing. Herein, it is shown that the introduction of a peptide to the side chain enables a continuous polydiacetylene fibre to exhibit ultrafast, reversible thermochromatic transitions at a high temperature of 200 °C. In particular, the reversible thermochromism occurs even at a temperature change rate of 5000 K s−1, much higher than the typical ∼1 K s−1 of other sensing polymers. The formation of a hierarchically assembled structure is responsible for this remarkable chromatism. In addition, a general and effective model is discovered to quantitatively predict the critical temperature of the colour change in polydiacetylene and guide the development of chromatic materials.

 Please wait while we load your content...
Please wait while we load your content...