Single-molecule spectroscopy of photosynthetic proteins in solution: exploration of structure–function relationships
Abstract
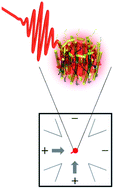
In photosynthetic light harvesting, absorbed photoenergy transfers through networks of pigment–protein complexes to a central location, known as the reaction center, for conversion to chemical energy. This process occurs with remarkable near-unity quantum efficiency. Pigment–protein complexes exhibit emergent properties very different from those of their component molecules, resulting from interactions among the pigments and between the pigments and the surrounding protein environment. Thus, the precise molecular mechanisms of the energy transport process are complex and coupled, obscuring their molecular origin. Furthermore, because these interactions are sensitive to the molecular structure, they vary from complex to complex and vary in time for each individual complex due to fluctuations of the protein's conformation. To explore the effect of complex-to-complex variation, we study individual photosynthetic proteins, one at a time. We use a solution-phase, single-molecule technique, known as the Anti-Brownian Electrokinetic (ABEL) trap, to elucidate the conformational dynamics of single photosynthetic pigment–protein complexes without introducing additional perturbations from immobilization strategies. After reviewing the principles of the ABEL trap, we demonstrate its application to the study of several photosynthetic pigment–protein complexes. We demonstrate that the ABEL trap approach can lead to an increased understanding of photosynthetic complexes by presenting three examples: (1) analysis of photodegradation pathways, (2) characterization of complex-to-complex heterogeneity, and (3) identification of distinct functional forms.

 Please wait while we load your content...
Please wait while we load your content...