Insight into aggregation-induced emission characteristics of red-emissive quinoline-malononitrile by cell tracking and real-time trypsin detection†
Abstract
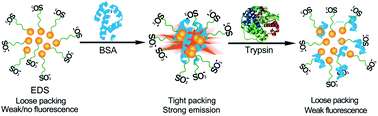
Water-soluble, long wavelength fluorescent aggregation-induced emission (AIE)-active materials are in great demand for high contrast biosensing and bioimaging. The substitution position effects of the sulfonate group on the basis of two quinoline-malononitrile (QM) derivatives (EDS and EDPS) provide insight into efficient modulation in the hydrophilicity, emitting color, and specific AIE characteristics. EDS shows a unique AIE behaviour in aqueous solution, but EDPS does not. The abnormal non-fluorescence aggregation for EDS in pure water is capsule-like with loose packing characteristics, but still has enough cavities or free volume to consume the radiative energy, resulting in nearly no fluorescence. When binding with the protein BSA, the sulfonate unit as a conformation function group (CFG) plays a vital role in altering its initial loose ensemble into tightly compact aggregation with light-up AIE characteristics. By cell tracking, dynamic light scattering (DLS) and transmission electron microscopy (TEM), the key role of sulfonate groups in the conformation alteration has been well demonstrated for the first time. Moreover, EDS is successfully exploited in a label-free real time AIE fluorescent assay for trypsin detection and inhibitor screening. The hydrophilic sulfonate group from the different substitution position in the AIE-active QM building blocks provides an effective way to tailor the intermolecular aggregation associated with molecular stacking, especially for in situ cell tracking and real-time trypsin detection.

 Please wait while we load your content...
Please wait while we load your content...