Asymmetric addition of chiral boron-ate complexes to cyclic iminium ions†
Abstract
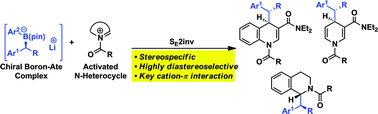
Boron-ate complexes derived from enantioenriched secondary benzylic boronic esters and aryl lithiums have been reacted with quinolinium, pyridinium and dihydroisoquinolinium salts to give enantioenriched heterocyclic structures with very high diastereocontrol over two contiguous stereogenic centres (87: 1 3–99 : 1 dr; >95 : 5 es). The salts were derived from the corresponding heterocycle and Troc-Cl or dimethylTroc-Cl. In the case of the quinolinium and pyridinium salts, the presence of a 3-carboxyamide group increased both reactivity and diastereoselectivity. The unusually high diastereoselectivity observed is thought to originate from strong cation–π interactions between the cationic heterocycle and the electron rich benzylic boronate complex with minimisation of steric interactions between the substituents on the ate complex and the non-planar substituents on the heterocycle.

 Please wait while we load your content...
Please wait while we load your content...