Palladium nanoparticles supported on ionic liquid modified, magnetic nanobeads – recyclable, high-capacity catalysts for alkene hydrogenation†
Abstract
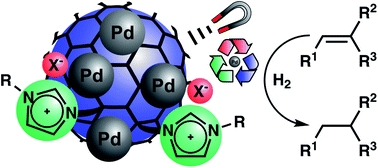
Magnetic hybrid materials have been synthesized as recyclable catalysts for alkene hydrogenation. The materials consist of magnetic nanobeads functionalized with imidazolium-based ionic liquids and optional polymer shells. Palladium nanoparticles (NPs) were synthesized on the surface of these supports by two different methods and evaluated as catalysts for alkene hydrogenation. Deposition of palladium(0) onto the magnetic nanobeads by microwave decomposition of Pd2(dba)3·CHCl3 leads to more efficient catalysts than the reduction of a Pd(II) precursor. Reactivity, recycling ability and ease of separation of the catalysts are compared. A hybrid material without polymer shells and a quite flexible ionic liquid was identified as the most promising for stabilizing Pd NPs resulting in a catalyst that shows high activity (TOF up to 330 h−1), good recycling ability, and minor metal leaching into the product. Notably, the activity of this catalyst increases with an enhanced Pd loading, contrasting related systems for which a decrease of activity is observed due to agglomeration. Therefore, this recyclable, high-capacity system is especially attractive for large-scale applications, requiring just a minimal amount of supporting material for the recycling of expensive Pd that is readily achieved by magnetic decantation.

 Please wait while we load your content...
Please wait while we load your content...