Hydrogen-bond-assisted isotactic-specific radical polymerization of N-vinyl-2-pyrrolidone with tartrate additives in toluene at low temperatures: high-resolution 1H NMR analysis†
Abstract
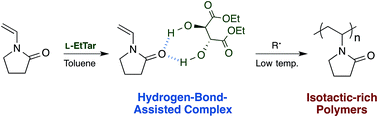
A diethyl L-tartrate (L-EtTar)-assisted radical polymerization of N-vinyl-2-pyrrolidone has been developed as the first reported example of the synthesis of isotactic-rich poly(N-vinyl-2-pyrrolidone) (PVP). The addition of L-EtTar in toluene at temperatures of −40 °C and lower led to a significant increase in the polymer yield by one order of magnitude compared with the reaction in the absence of L-EtTar. Decreasing the polymerization temperature led to increases in the isotacticity of the PVP, with the mm triad reaching 66.4% at −93 °C. 1H NMR measurement at 920 MHz was conducted to establish a reliable strategy for quantifying the triad tacticities. High-temperature NMR measurements at 250 °C were performed using a specially-designed NMR probe, which led to dramatic narrowing of the 1H line width.

 Please wait while we load your content...
Please wait while we load your content...