Charge density distribution and electrostatic interactions of ethionamide: an inhibitor of the enoyl acyl carrier protein reductase (inhA) enzyme of Mycobacterium tuberculosis†
Abstract
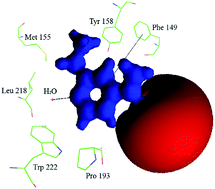
An experimental charge density analysis of an anti-TB drug ethionamide was carried out from high resolution X-ray diffraction at 100 K to understand its charge density distribution and electrostatic properties. The experimental results were validated from periodic theoretical charge density calculations performed using CRYSTAL09 at the B3LYP/6-31G** level of theory. The electron density ρbcp(r) and the Laplacian of electron density ∇2ρbcp(r) of the molecule calculated from both the methods display the charge density distribution of the ethionamide molecule in the crystal field. The electrostatic potential map shows a large electropositive region around the pyridine ring and a large electronegative region at the vicinity of the thiol atom. The calculated experimental dipole moment is 10.6D, which is higher than the value calculated from theory (8.2D). The topological properties of C–H⋯S, N–H⋯N and N–H⋯S hydrogen bonds were calculated, revealing their strength. The charge density analysis of the ethionamide molecule determined from both the experiment and theory gives the topological and electrostatic properties of the molecule, which allows to precisely understand the nature of intra and intermolecular interactions.

 Please wait while we load your content...
Please wait while we load your content...