Nanoassembly of an amphiphilic cyclodextrin and Zn(ii)-phthalocyanine with the potential for photodynamic therapy of cancer†
Abstract
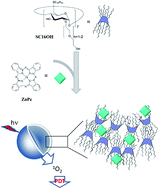
Due to their poor solubility and propensity to aggregate in aqueous media, therapeutic application of several photosensiting agents, such as phthalocyanines, in photodynamic therapy (PDT) of solid tumors is severely hampered. With the aim to propose a novel nanotechnological approach, in this paper biodegradable nanoassemblies based on heptakis (2-oligo(ethyleneoxide)-6-hexadecylthio-)-β-CD (SC16OH) and zinc-phthalocyanine (ZnPc) were developed and tested. Nanoassemblies, prepared by the emulsion–solvent evaporation technique, displayed a hydrodynamic diameter around 200 nm, a negative zeta potential and a satisfactory entrapment efficiency of ZnPc. Steady-state and time resolved fluorescence emission spectroscopy studies showed the entrapment of ZnPc as a monomer in the carrier, with a low tendency to self-aggregate and consequently a fairly good propensity to generate singlet oxygen after photoactivation. The interaction of ZnPc with SC16OH was elucidated by 1H-NMR, which suggested the formation of complexes between drug and both hydrophobic and hydrophilic moieties of the amphiphile. Finally, in vitro potential of the nanoassembly was evaluated in HeLa cells by following cellular uptake and photobiological activity. Overall, results suggest the suitability of the nanoassembly based on SC16OH for delivering ZnPc to cancer cells, thus inducing photodynamic anticancer effects.

 Please wait while we load your content...
Please wait while we load your content...