High adsorption and efficient visible-light-photodegradation for cationic Rhodamine B with microspheric BiOI photocatalyst†
Abstract
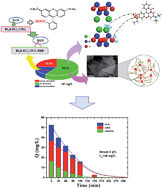
BiOI microspheres were synthesized using an EG-assisted one-pot solvothermal method. The morphology, structure, surface area, pore-size distribution and surface electrical property of the BiOI photocatalyst were characterized using XRD, SEM, TEM, BET and zeta potential measurements. The results showed that the microspheric structure consisting of numerous nanosheets manifested a large BET area 59.3 m2 g−1 with a mesoporous structure of 14.1 nm. Besides, its surfaces were negatively charged in general. Adsorption kinetics and isotherm analysis indicated that the adsorption of cationic Rhodamine B (RhB) onto BiOI followed the pseudo-second-order model, and the Freundlich equation sufficiently described the sorption isotherm. As the pH increased, the equilibrium adsorption concentration exhibited a downward trend. The adsorption mechanism was tentatively proposed using FTIR and XPS spectral analysis. The three main functional mechanisms involved in the adsorption process were aperture adsorption, electrostatic interaction and chemical coordination effect. BiOI exhibited high photodegradation efficiency for highly concentrated RhB solution and adsorption enhanced photocatalytic efficiency by improving the utilization of reactive species in the photocatalytic oxidation process.

 Please wait while we load your content...
Please wait while we load your content...