In situ synthesis of flowery-shaped α-FeOOH/Fe2O3 nanoparticles and their phase dependent supercapacitive behaviour†
Abstract
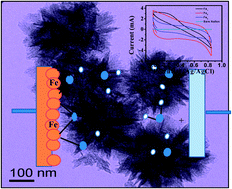
In situ, one-step, facile synthetic strategies for flowery-shaped iron oxide nanoparticles were developed. Herein, we report simplified controlled synthesis of 2-line ferrihydrite–goethite core–shell particles for the first time in a semi-aqueous-organic medium. The present route offered phase selectivity by controlling only the aqueous-to-organic phase ratio. The synthesised nanoparticles have high surface areas of 110 m2 g−1 and 185 m2 g−1 for 2-line ferrihydrite and core–shell goethite, respectively. Further, flowery-shaped hematite nanoparticles were obtained by annealing core–shell iron oxide nanoparticles at 400 °C. Phase purities were confirmed by XRD (X-ray diffraction), IR (infrared spectroscopy), and XPS (X-ray photo electron spectroscopy) analysis. Formation of the core–shell nanostructure for the iron oxide samples was confirmed by Mössbauer and selected area electron diffraction (SAED) studies. All the synthesized iron oxide materials were studied for their supercapacitor behaviour by cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), and chrono-potentiometry charge–discharge measurements. Specific capacitances for core–shell α-FeOOH and α-Fe2O3 were found to be 160 F g−1 and 200 F g−1, respectively. These values were much higher as compared to the previous reported values for pure phases of iron oxides. The chrono-potentiometric charge–discharge study for all the three samples revealed their self-discharging capacities. Moreover, these iron oxide composite electrodes exhibited excellent cycling performance with >99% capacitance retention over 500 cycles. Electrochemical performance of the two-electrode system was also studied. Furthermore, the electrochemical impedance spectroscopy (EIS) demonstrated that the electrochemical resistance of α-Fe2O3 was slightly reduced with the number of cycles, indicating easier access for intercalation/deintercalation of charges in the flowery-structured materials. Thus, the present material can be used as an electrochemical supercapacitor for high-performance energy storage devices in future.

 Please wait while we load your content...
Please wait while we load your content...