Direct alkenyl C–H functionalization of cyclic enamines with carboxylic acids via Rh catalysis assisted by hydrogen bonding†‡
Abstract
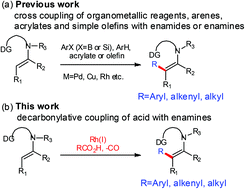
Enamines and enamides are important synthetic intermediates. The transition metal catalyzed C–C coupling through direct β-C–H activation of enamines or enamides is an important method for their functionalization. But so far the effective coupling partners have been limited to organometallic reagents, arenes, olefins, and acrylates. In this study, a highly efficient method was developed to use carboxylic acids, an easily available and cheap carbon source, as coupling partners for the direct β-C–H functionalization of enamines in the presence of the Rh(I) catalyst and the aminopyridinyl directing group through decarbonylation coupling. The reaction was proved to be assisted by hydrogen bonding. The directing group was easily removed under acidic conditions. This method provides a useful alternative approach to synthesize C-alkylated and arylated cyclic diketones.

 Please wait while we load your content...
Please wait while we load your content...