Injectable hydrogels with in situ-forming hydrophobic domains: oligo(d,l-lactide) modified poly(oligoethylene glycol methacrylate) hydrogels†
Abstract
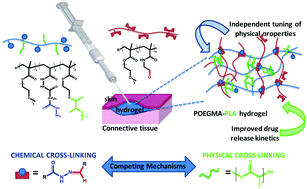
Injectable, in situ-gelling nanostructured hydrogels have been prepared from hydrazide and aldehyde-functionalized polymer precursors based on a copolymer of oligo(ethylene glycol) methacrylate (OEGMA) and an oligo(lactic acid) macromonomer (OLA) with varying lactic acid chain lengths. The resulting hydrogels contain a mix of chemical (hydrazone bond formation between hydrazide and aldehyde groups) and physical (hydrophobic interactions between OLA chains) cross-links which form competitively as a function of the OLA chain length and density. An increase in the OLA chain length and density results in the formation of more physical cross-links and fewer chemical cross-links. Tuning the relative prevalence of physical and chemical cross-link formation facilitated largely independent tuning of gel mechanics relative to gel swelling and degradation. Small-angle neutron scattering of these OLA-containing hydrogels reveals a microstructure consisting of associative hydrophobic domains, based on an increased scattering intensity and decreased blob size relative to that observed for POEGMA hydrogels prepared without the OLA co-monomer. The presence of hydrophobic OLA domains increases the uptake and slows the release of bovine serum albumin, a protein well-known to associate with hydrophobic domains. Coupled with the observed cytocompatibility of the reactive precursor polymers used to prepare the hydrogels, we anticipate significant potential applications of these hydrogels for the prolonged release of hydrophobic cargoes.

 Please wait while we load your content...
Please wait while we load your content...