Chemical functionalization of emulsion-templated porous polymers by thiol–ene “click” chemistry
Abstract
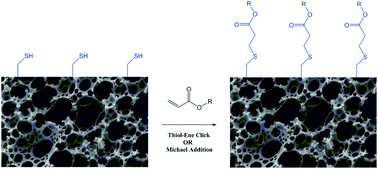
Highly porous polymers (polyHIPEs) have been prepared by the photopolymerization of high internal phase emulsions (HIPEs) with varying ratios of thiol and acrylate monomers. The resulting polymers have a nominal porosity of 80%, and are seen to have a well-defined, interconnected pore morphology, with average pore diameters ranging from 30 to 60 μm. The polyHIPE polymers have been shown using a colourimetric (Ellman's) assay to contain residual thiols which are reactive towards a range of (meth)acrylates (hexafluoroisopropyl acrylate, fluorescein O-acrylate and poly(ethylene glycol) methyl ether methacrylate). Functionalization was explored using thermally- and UV-initiated radical-mediated “click” reactions and an amine-catalysed Michael addition reaction. The extent of functionalization was investigated qualitatively and quantitatively using a range of techniques (solid state NMR spectroscopy; FTIR spectroscopy; X-ray photoelectron spectroscopy (XPS); observation of fluorescence); high levels of conversion (up to 90–95%) were observed for the thermally-initiated radical reaction and the Michael reaction.

 Please wait while we load your content...
Please wait while we load your content...