On the effect of using RAFT and FRP for the bulk synthesis of acrylic and methacrylic molecularly imprinted polymers†
Abstract
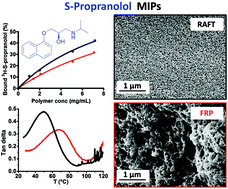
Molecularly imprinted polymers (MIPs) are synthetic polymeric receptors, capable of specifically binding a target molecule, just like a biological antibody. There has been a recent trend to improve the properties of these materials by using modern methods of controlled radical polymerization (CRPs) for their synthesis. Despite the recognized advantages associated with their “living character”, the effect of the “controlled nature” has still to be clearly demonstrated. This is far from obvious as the high amounts of short cross-linkers normally used for their synthesis complicate the formation of homogeneous polymer networks. In order to gain more insights into the potential benefits for the binding properties of MIPs resulting from the use of CRPs, the imprinting of a model target (S-propranolol) has been used to compare reversible addition–fragmentation chain transfer polymerisation (RAFT) and free-radical polymerisation (FRP) on acrylic and methacrylic matrices. While most MIPs are based on methacrylates, we used acrylates as a “difficult imprinting matrix” for comparison. In fact, the absence of the methyl groups in their polymer back-bone reduces their entanglement, resulting in a more flexible network. This renders the material more difficult to imprint, and at the same time makes it easier to evaluate the effects of RAFT polymerization and FRP on structural parameters and thus binding properties. Moreover, we also progressively reduced the amount of cross-linking in order to explore the effects of RAFT and FRP on a wider range of scaffold rigidities. Although MIPs are normally highly cross-linked, some recent emerging applications require lower degrees of cross-linking. Binding experiments, SEM, BET, DMA, swelling and nanoindentation analyses revealed that RAFT is effective in promoting the synthesis of more homogeneous networks compared to FRP, even at very high cross-linker contents, which results in higher target affinities, especially in the case of acrylates.

 Please wait while we load your content...
Please wait while we load your content...