Kinetic analysis of nitroxide radical formation under oxygenated photolysis: toward quantitative singlet oxygen topology†
Abstract
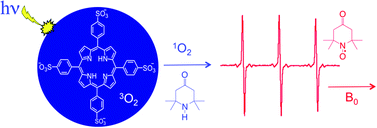
Reaction kinetics for two sterically hindered secondary amines with singlet oxygen have been studied in detail. A water soluble porphyrin sensitizer, 5,10,15,20-tetrakis-(4-sulfunatophenyl)-21,23H-porphyrin (TPPS), was irradiated in oxygenated aqueous solutions containing either 2,2,6,6-tetramethylpiperidin-4-one (TMPD) or 4-[N,N,N-trimethyl-ammonium]-2,2,6,6-tetramethylpiperidinyl chloride (N-TMPCl). The resulting sensitization reaction produced singlet oxygen in high yield, ultimately leading to the formation of the corresponding nitroxide free radicals (R2NO) which were detected using steady-state electron paramagnetic resonance (EPR) spectroscopy. Careful actinometry and EPR calibration curves, coupled with a detailed kinetic analysis, led to a simple and compact expression relating the nitroxide quantum yield ΦR2NO (from the doubly-integrated EPR signal intensity) to the initial amine concentration [R2NH]i. With all other parameters held constant, a plot of ΦR2NOvs. [R2NH]i gave a straight line with a slope proportional to the rate constant for nitroxide formation, kR2NO. This establishment of a rigorous quantitative relationship between the EPR signal and the rate constant provides a mechanism for quantifying singlet oxygen production as a function of its topology in heterogeneous media. Implications for in vivo assessment of singlet oxygen topology are briefly discussed.

 Please wait while we load your content...
Please wait while we load your content...