A new class of high-contrast Fe(ii) selective fluorescent probes based on spirocyclized scaffolds for visualization of intracellular labile iron delivered by transferrin†
Abstract
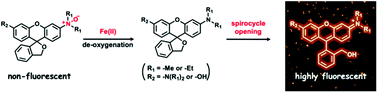
Iron is an essential metal nutrient that plays physiologically and pathologically important roles in biological systems. However, studies on the trafficking, storage, and functions of iron itself in living samples have remained challenging due to the lack of efficient methods for monitoring labile intracellular iron. Herein, we report a new class of Fe2+-selective fluorescent probes based on the spirocyclization of hydroxymethylrhodamine and hydroxymethylrhodol scaffolds controlled by using our recently established N-oxide chemistry as a Fe2+-selective switch of fluorescence response. By suppressing the background signal, the spirocyclization strategy improved the turn-on rate dramatically, and reducing the size of the substituents of the N-oxide group enhanced the reaction rate against Fe2+, compared with the first generation N-oxide based Fe2+ probe, RhoNox-1. These new probes showed significant enhancements in the fluorescence signal against not only the exogenously loaded Fe2+ but also the endogenous Fe2+ levels. Furthermore, we succeeded in monitoring the accumulation of labile iron in the lysosome induced by transferrin-mediated endocytosis with a turn-on fluorescence response.

 Please wait while we load your content...
Please wait while we load your content...