Triflic acid-promoted cycloisomerization of 2-alkynylphenyl isothiocyanates and isocyanates: a novel synthetic method for a variety of indole derivatives†
Abstract
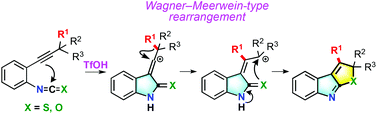
A new approach towards the synthesis of indole derivatives via triflic acid-promoted cycloisomerization with rearrangement of 2-(alkyn-1-yl)phenyl isothiocyanates and 2-(alkyn-1-yl)phenyl isocyanates has been achieved. By this methodology, structurally diverse types of indole derivatives such as thieno- and furo-indoles, spiro-indolethiones, spiro-oxindoles, and 3-alkylidene-oxindoles were synthesized.

 Please wait while we load your content...
Please wait while we load your content...