Electrodeposited polyethylenedioxythiophene with infiltrated gel electrolyte interface: a close contest of an all-solid-state supercapacitor with its liquid-state counterpart†
Abstract
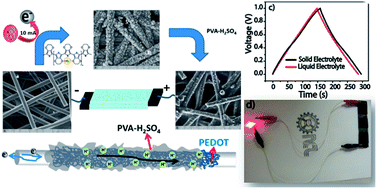
We report the design of an all-solid-state supercapacitor, which has charge storage characteristics closely matching that of its liquid-state counterpart even under extreme temperature and humidity conditions. The prototype is made by electro-depositing polyethylenedioxythiophene (PEDOT) onto the individual carbon fibers of a porous carbon substrate followed by intercalating the matrix with polyvinyl alcohol–sulphuric acid (PVA–H2SO4) gel electrolyte. The electrodeposited layer of PEDOT maintained a flower-like growth pattern along the threads of each carbon fiber. This morphology and the alignment of PEDOT led to an enhanced surface area and electrical conductivity, and the pores in the system enabled effective intercalation of the polymer–gel electrolyte. Thus, the established electrode–electrolyte interface nearly mimics that of its counterpart based on the liquid electrolyte. Consequently, the solid device attained very low internal resistance (1.1 Ω cm−2) and a high specific capacitance (181 F g−1) for PEDOT at a discharge current density of 0.5 A g−1. Even with a high areal capacitance of 836 mF cm−2 and volumetric capacitance of 28 F cm−3, the solid device retained a mass-specific capacitance of 111 F g−1 for PEDOT. This is in close agreement with the value displayed by the corresponding liquid-state system (112 F g−1), which was fabricated by replacing the gel electrolyte with 0.5 M H2SO4. The device also showed excellent charge–discharge stability for 12 000 cycles at 5 A g−1. The performance of the device was consistent even under wide-ranging humidity (30–80%) and temperature (−10 to 80 °C) conditions. Finally, a device fabricated by increasing the electrode area four times was used to light an LED, which validated the scalability of the process.

 Please wait while we load your content...
Please wait while we load your content...