Controlled colloidal synthesis of iron pyrite FeS2 nanorods and quasi-cubic nanocrystal agglomerates
Abstract
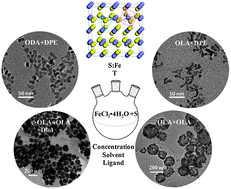
Earth-abundant and nontoxic pyrite iron disulfide (FeS2) is very promising for photovoltaic applications but the phase purity and the morphology of iron pyrite nanocrystals (NCs) have a significant impact on the solar cell performance. In this work, we systematically investigated reaction conditions and the local chemical environment on the phase purity and morphology of iron pyrite NCs synthesized via the hot injection method. By using different solvents to dissolve iron and sulfur agents, varying reactant concentrations, and adding trioctylphosphine oxide (TOPO) or 1,2-hexadecanediol (Diol) into the reaction solution, iron pyrite short, branched and chromosome-like rods were obtained with a diameter of ∼10 nm and a length of ∼20–30 nm as well as quasi-cubic NC agglomerates with a size of ∼200 nm. Our experimental results show that the molar ratio of sulfur to iron and the reaction temperature are two critical factors in determining the crystalline phase of the synthesized materials. A mechanism involving the generation of H2S is proposed to explain the phase purity observed. The as-synthesized iron pyrite NCs can be dispersed well in chloroform, chlorobenzene, toluene, and hexane and thus are promising in solution-processable photovoltaic applications.

 Please wait while we load your content...
Please wait while we load your content...