In vivo photoacoustic molecular imaging of breast carcinoma with folate receptor-targeted indocyanine green nanoprobes
Abstract
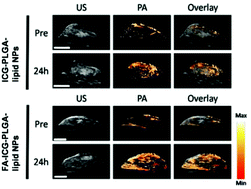
As an optical-acoustic hybrid imaging technology, photoacoustic imaging uniquely combines the advantages of rich optical contrast with high ultrasonic resolution in depth, opening up many new possibilities not attainable with conventional pure optical imaging technologies. To perform photoacoustic molecular imaging, optically absorbing exogenous contrast agents are needed to enhance the signals from specifically targeted disease activity. In this work, we designed and developed folate receptor targeted, indocyanine green dye doped poly(D,L-lactide-co-glycolide) lipid nanoparticles (FA-ICG-PLGA-lipid NPs) for molecular photoacoustic imaging of tumor. The fabricated FA-ICG-PLGA-lipid NPs exhibited good aqueous stability, a high folate-receptor targeting efficiency, and remarkable optical absorption in near-infrared wavelengths, providing excellent photoacoustic signals in vitro. Furthermore, after intravenous administration of FA-ICG-PLGA-lipid NPs, mice bearing MCF-7 breast carcinomas showed significantly enhanced photoacoustic signals in vivo in the tumor regions, compared with those using non-targeted ICG-PLGA-lipid NPs. Given the existing wide clinical use of ICG and PLGA, the developed FA-ICG-PLGA-lipid NPs, in conjunction with photoacoustic imaging technology, offer a great potential to be translated into the clinic for non-ionizing molecular imaging of breast cancer in vivo.

 Please wait while we load your content...
Please wait while we load your content...