Propane dehydrogenation over Pt–Cu bimetallic catalysts: the nature of coke deposition and the role of copper
Abstract
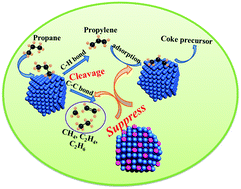
This paper describes an investigation of the promotional effect of Cu on the catalytic performance of Pt/Al2O3 catalysts for propane dehydrogenation. We have shown that Pt/Al2O3 catalysts possess higher propylene selectivity and lower deactivation rate as well as enhanced anti-coking ability upon Cu addition. The optimized loading content of Cu is 0.5 wt%, which increases the propylene selectivity to 90.8% with a propylene yield of 36.5%. The origin of the enhanced catalytic performance and anti-coking ability of the Pt–Cu/Al2O3 catalyst is ascribed to the intimate interaction between Pt and Cu, which is confirmed by the change of particle morphology and atomic electronic environment of the catalyst. The Pt–Cu interaction inhibits propylene adsorption and elevates the energy barrier of C–C bond rupture. The inhibited propylene adsorption diminishes the possibility of coke formation and suppresses the cracking reaction towards the formation of lighter hydrocarbons on Pt–Cu/Al2O3, while a higher energy barrier for C–C bond cleavage suppresses the methane formation.

 Please wait while we load your content...
Please wait while we load your content...