Synthesis and electrocatalytic properties of PtBi nanoplatelets and PdBi nanowires†
Abstract
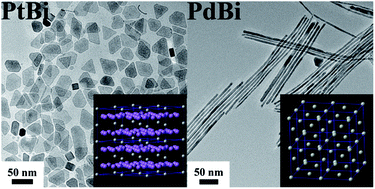
We have demonstrated a one-pot, facile and rapid strategy to synthesize novel PtBi nanoplatelets (NPLs) and PdBi nanowires (NWs) with controlled shape, size, and composition in the presence of oleylamine (OAm) and NH4Br. In contrast to the conventional face centered cubic (fcc) structure of Pt-based NPs, PtBi possesses a chemically ordered intermetallic hexagonal close packed (hcp) structure. Using this uniaxial crystal structural character of PtBi, we succeed in synthesizing two-dimensional (2-D) PtBi NPLs. Significantly, the electrochemical studies indicate that the as-prepared 2-D PtBi NPLs exhibit enhanced electrocatalytic activity toward formic acid and methanol oxidation with larger oxidation current density, higher tolerance to CO poisoning, and more negative onset potential in comparison with the commercial Pt/C catalyst. This is attributed to the addition of second metal Bi. In addition, to the best of our knowledge, this is the first time that synthesis of one-dimensional (1-D) PdBi alloy NWs has been reported. The as-synthesized 1-D PdBi bimetallic NWs may find promising potential applications in various fields, such as fuel cells, electrochemical sensors, and organocatalysis.

 Please wait while we load your content...
Please wait while we load your content...